Pharmaceuticals
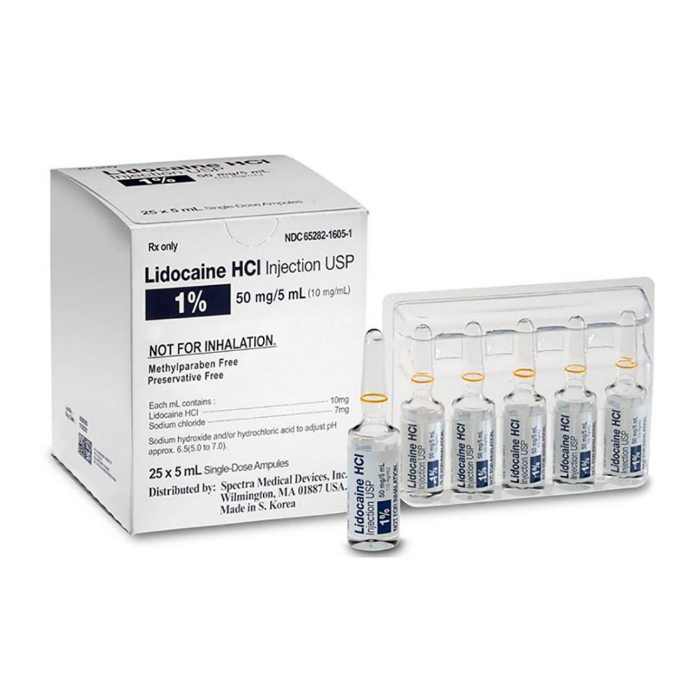
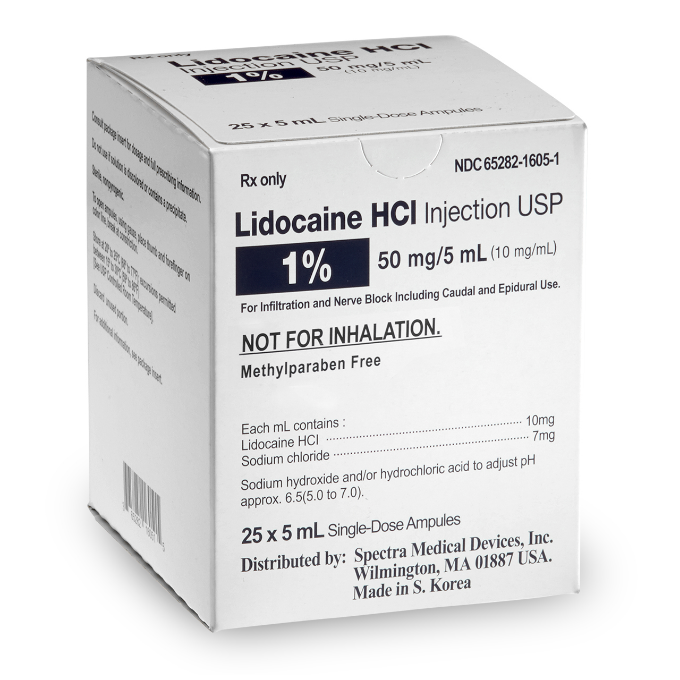
Lidocaine Hydrochloride Injection, USP, 1%, 5 mL Single-Dose Ampules
Spectra offers Lidocaine Hydrochloride Injection, USP, 1%, indicated for production of local or regional anesthesia by infiltration techniques such as percutaneous injection and intravenous regional anesthesia, by peripheral nerve block techniques such as brachial plexus and intercostal and by central neural techniques such as lumbar and caudal epidural blocks.
Features:
- Lidocaine Hydrochloride Injection, USP, 1%
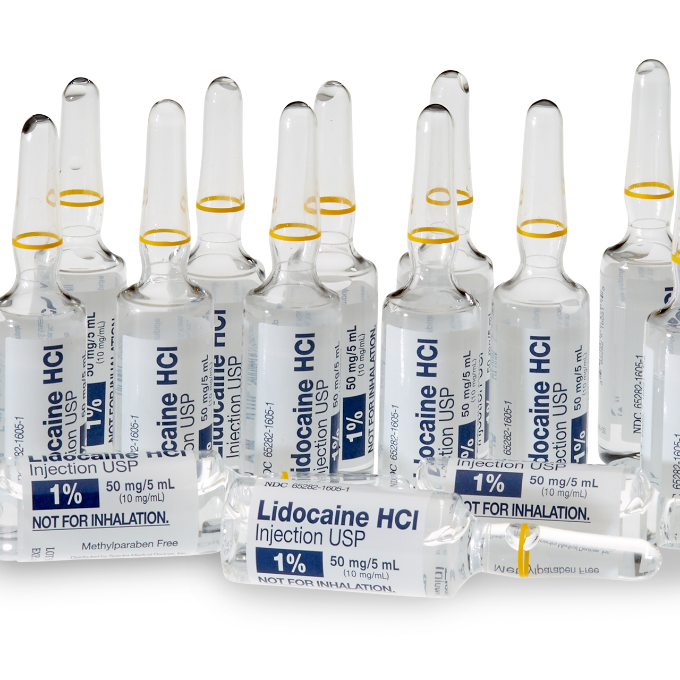
- Glass Ampules
- Preservative Free
How to Order:
- 5 mL Single-Dose Glass Ampules*
- Box of 25 / Carton of 900
- NDC: 65282-1605-1
*USP Type I Glass Ampules, hermetically sealed, compatible with EtO sterilization. Glass is impermeable to EtO per ANSI/AAMI ST41:1999 Standard
> Important Safety Information
To place an order, request samples or information: